Once physicists started accelerating particles to high energies in the 1930s, it didn’t take them long to think of a killer app for this new technology: zapping tumors.
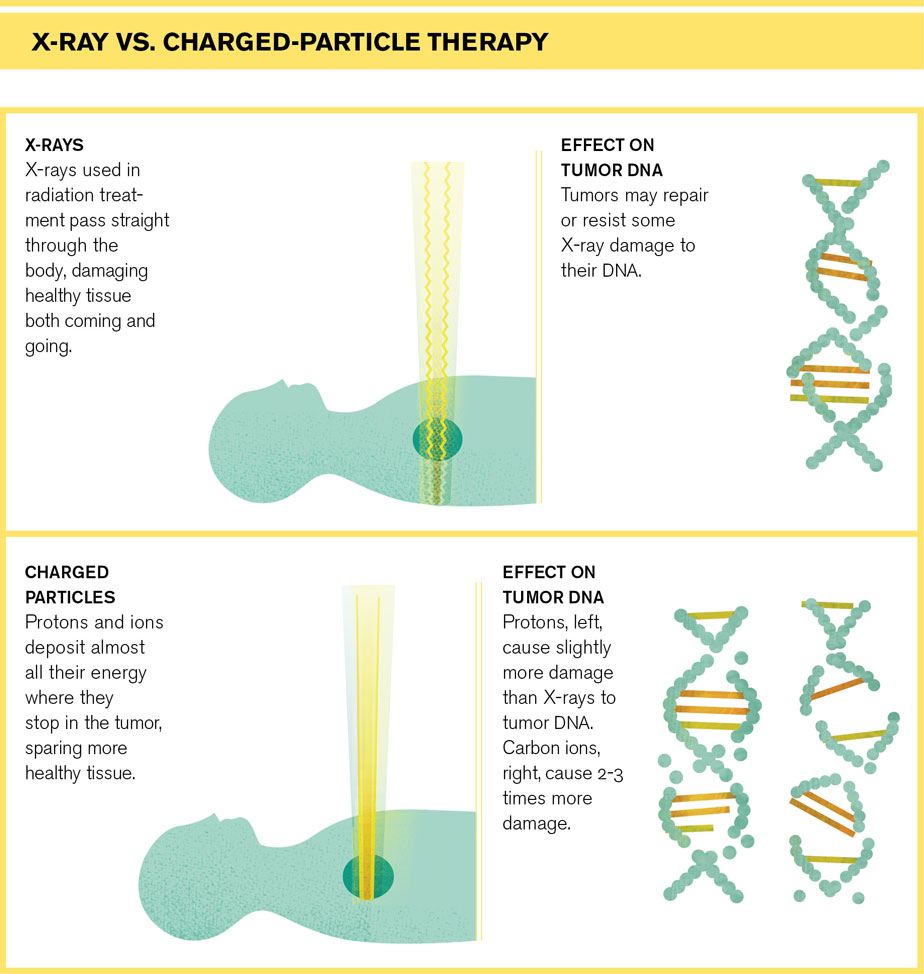
Standard radiation treatments, which had already been around for decades, send X-rays straight through the tumor and out the other side of the body, damaging healthy tissue both coming and going. But protons and ions—atoms stripped of electrons—slow when they hit the body and come to a stop, depositing most of their destructive energy at their stopping point. If you tune a beam of protons or ions so they stop inside a tumor, you can deliver the maximum dose of radiation while sparing healthy tissue and minimizing side effects. This makes it ideal for treating children, whose developing bodies are particularly sensitive to radiation damage, and for cancers very close to vital tissues such as the optic nerves or spinal cord.
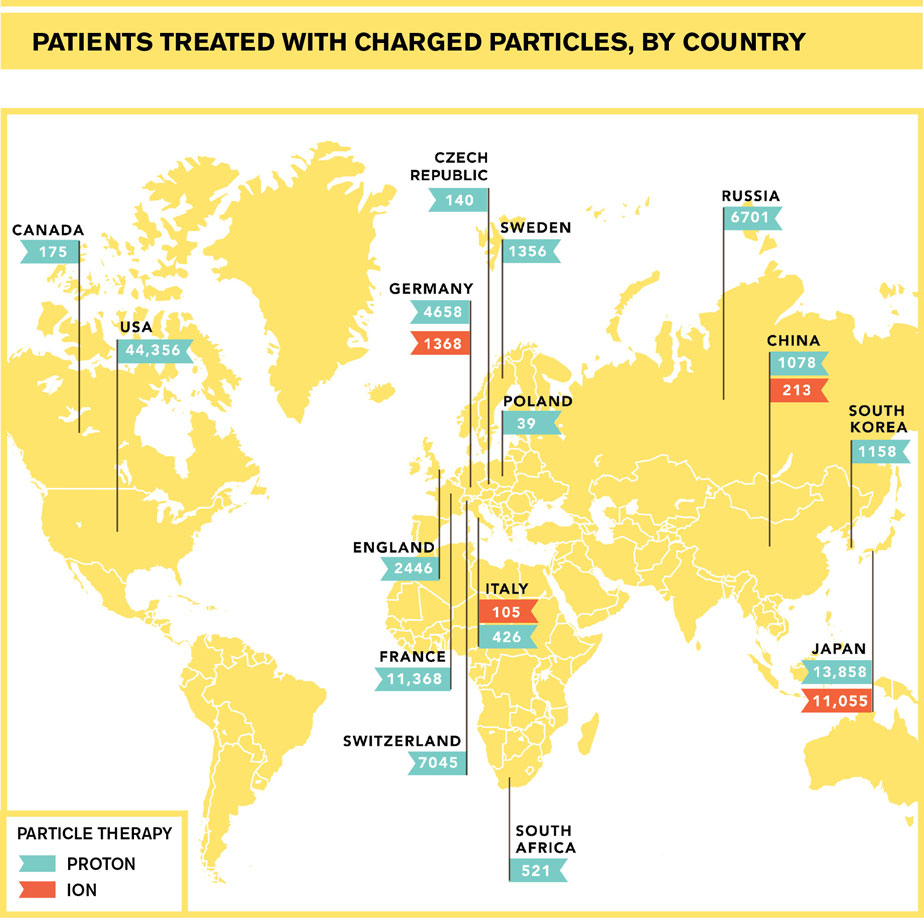
Today, nearly 70 years after American particle physicist Robert Wilson came up with the idea, proton therapy has been gaining traction worldwide and in the United States, where 14 centers are treating patients and nine more are under construction. Ions such as carbon, helium and oxygen are being used to treat patients in Germany, Italy, China and Japan. More than 120,000 patients had been treated with various forms of charged-particle therapy by the end of 2013, according to the Particle Therapy Co-Operative Group.
New initiatives from CERN research center in Europe and the Department of Energy and National Cancer Institute in the United States are aimed at moving the technology along, assessing its strengths and limitations and making it more affordable.
And physicists are still deeply involved. No one knows more about building and operating particle accelerators and detectors. But there’s a lot more to know. So they’ve been joining forces with physicians, engineers, biologists, computer scientists and other experts to make the equipment smaller, lighter, cheaper and more efficient and to improve the way treatments are done.
“As you get closer to the patient, you leave the world accelerator physicists live in and get closer to the land of people who have PhDs in medical physics,” says Stephen Peggs, an accelerator physicist at Brookhaven National Laboratory.
“It’s alignment, robots and patient ergonomics, which require just the right skill sets, which is why it’s fun, of course, and one reason why it’s interesting—designing with patients in mind.”
Knowing where to stop
The collaborations that make charged-particle therapy work go back a long way. The first experimental treatments took place in 1954 at what is now Lawrence Berkeley National Laboratory. Later scientists at Fermi National Accelerator Laboratory designed and built the circular accelerator at the heart of the first hospital-based proton therapy center in the United States, opened in 1990 at California’s Loma Linda University Medical Center.
A number of private companies have jumped into the field, opening treatment centers, selling equipment and developing more compact and efficient treatment systems that are designed to cut costs. ProTom International, for instance, recently received US Food and Drug Administration approval for a system that’s small enough and light enough to ship on a plane and move in through a door, so it will no longer be necessary to build the treatment center around it. Other players include ProCure, Mevion, IBA, Varian Medical Systems, ProNova, Hitachi, Sumitomo and Mitsubishi.
The goal of any treatment scheme is to get the beam to stop in exactly the right spot; the most advanced systems scan a beam back and forth to “paint” the 3-D volume of the tumor with great precision. Aiming it is not easy, though. Not only is every patient’s body different—a unique conglomeration of organs and tissues of varying densities—but every patient breathes, so the target is in constant motion.
Doctors use X-ray CT scans—the CT stands for “computed tomography”—to make a 3-D image of the tumor and its surroundings so they can calculate the ideal stopping point for the proton beam. But since protons don’t travel through the body exactly the same way X-rays do—their paths are shifted by tiny, rapid changes in the tissues they encounter along the way—their end points can differ slightly from the predicted ones.
Physicists are trying to reduce that margin of error with a technology called proton CT.
Reconnoitering with proton CT
The idea is simple: Use protons rather than X-rays to make the images. The protons are tuned to high enough energies that they go through the body without stopping, depositing about one-tenth as much radiation along their path as X-rays do.
Detectors in front of and behind the body pinpoint where each proton beam enters and leaves, and a separate detector measures how much energy the protons lose as they pass through tissues. By directing proton beams through the patient from different angles, doctors can create a 3-D image that tells them, much more accurately than X-rays, how to tune the proton beam so it stops inside the tumor.
Two teams are now in friendly competition, testing rival ways to perform proton CT on “phantom” human heads made of plastic. Both approaches are based on detectors that are staples in particle physics.
One team is made up of researchers from Northern Illinois University, Fermilab, Argonne National Laboratory and the University of Delhi in India and funded by the US Army Medical Research Acquisition Center in Maryland. They use a pair of fiber trackers on each side of the phantom head to pinpoint where the proton beams enter and exit. Each tracker contains thousands of thin plastic fibers. When a proton hits a fiber, it gives off a flash of light that is picked up by another physics standby—a silicon photomultiplier—and conveyed to a detector.
The team is testing this system, which includes computers and software for turning the data into images, at the CDH Proton Center in Warrenville, Illinois.
“The point is to demonstrate you can get the image quality you need to target the treatment more accurately with a lower radiation dose level than with X-ray CT,” says Peter Wilson, principal investigator for the Fermilab part of the project.
The second project, a collaboration between researchers at Loma Linda, University of California, Santa Cruz, and Baylor University, is fi-nanced by a $2 million grant from the National Institutes of Health. Their proton CT system is based on silicon strip detectors the Santa Cruz group developed for the Fermi Gamma-ray Space Telescope and the ATLAS experiment at CERN, among others. It’s being tested at Loma Linda.
“We know how to detect charged particles with silicon detectors. Charged particles for us are duck soup,” says UCSC particle physicist Hartmut Sadrozinski, who has been working with these detectors for more than 30 years. Since a single scan requires tracking about a billion protons, the researchers also introduced software packages developed for high-energy physics to analyze the high volume of data coming into the detector.
Proton CT will have to get a lot faster before it’s ready for the treatment room. In experiments with the phantom head, the system can detect a million protons per second, completing a scan in about 10 minutes, Sadrozinski says; the goal is to bring that down to 2 to 3 minutes, reducing the time the patient has to hold still and ensuring accurate images and dose delivery.
Trimming the size and cost of ion therapy
The first ion therapy center opened in Japan in 1994; by the end of 2013 centers in Japan, China, Germany and Italy had treated nearly 13,000 patients.
There’s reason to think ions could be more effective than protons or X-rays for treating certain types of cancer, according to a recent review of the field published in Radiation Oncology by researchers from the National Cancer Institute and Walter Reed National Military Medical Center. Ions deliver a more powerful punch than protons, causing more damage to a tumor’s DNA, and patient treatments have shown promise.
But the high cost of building and operating treatment centers has held the technology back, the researchers wrote; and long-term research on possible side effects, including the possibility of triggering secondary cancers, is lacking.
The cost of building ion treatment centers is higher in part because the ions are so much heavier than protons. You need bigger magnets to steer them around an accelerator, and heavier equipment to deliver them to the patient.
Two projects at Brookhaven National Laboratory aim to bring the size and cost of the equipment down.
One team, led by accelerator physicist Dejan Trbojevic, has developed and patented a simpler, less expensive gantry that rotates around a stationary patient to aim an ion beam at a tumor from various angles. Gantries for ion therapy can be huge—the one in use at the Heidelberg Ion-Beam Therapy Center in Germany weighs 670 tons and is tall as a jetliner. The new design shrinks the size of the gantry by making a single set of simpler, smaller magnets do double duty, both bending and focusing the particle beam.
In the second project, Brookhaven scientists are working with a Virginia company, Best Medical International, to design a system for treating patients with protons, carbon ions and other ion beams. Called the ion Rapidly Cycling Medical Synchrotron (iRCMS), it is designed to deliver ions to patients in smaller, more rapid pulses. With smaller pulses, the diameter of the beam also shrinks, along with the size of the magnets used to steer it. Brookhaven is building one of the system’s three magnet girders, radio-frequency acceleration cavities and a power supply for a prototype system. The end product must be simple and reliable enough for trained hospital technicians to operate for years.
“A particle accelerator for cancer treatment has to be industrial, robust—not the high-tech, high-performance, typical machine we’re used to,” says Brookhaven’s Peggs, one of the lead scientists on the project. “It’s more like a Nissan than a Ferrari.”
Launching a CERN initiative for cancer treatment
CERN, the international particle physics center in Geneva, is best known to many as the place where the Higgs boson was discovered in 2012. In 1996 it began collaborating on a study called PIMMS that designed a system for delivering both proton and ion treatments. That system evolved into the equipment at the heart of two ion therapy centers: CNAO, the National Center for Oncological Treatment in Pavia, Italy, which treated its first patient in 2011, and MedAustron, scheduled to open in Austria in 2015.
Now scientists at CERN want to spearhead an international collaboration to design a new, more compact treatment system that will incorporate the latest particle physics technologies. It’s part of a larger CERN initiative launched late last year with a goal of contributing to a global system for treating cancer with charged-particle beams.
Part of an existing CERN accelerator, the Low Energy Ion Ring, will be converted into a facility to provide various types of charged-particle beams for research into how they affect healthy and cancerous tissue. The lab will also consider developing detectors for making medical images and controlling the treatment beam, investigating ways to control the dose the patient receives and adapting large-scale computing for medical applications.
CERN will provide seed funding and seek out other funding from foundations, philanthropists and other sources, such as the European Union.
“Part of CERN’s mission is knowledge transfer,” says Steve Myers, director of the medical initiative, who spent the past five years running the Large Hadron Collider as director of accelerators and technology for CERN.
“We would like to make the technologies we have developed for particle physics available to other fields of research simply because we think it’s a nice thing to do,” he says. “All the things we do are related to the same goal, which is treating cancer tumors in the most effective and efficient way possible.”
Expanding the options in the US
In the US, the biggest barrier to setting up ion treatment centers is financial: Treatment centers cost hundreds of millions of dollars. Unlike in Europe and Asia, no government funding is available, so these projects have to attract private investors. But without rigorous studies showing that ion therapy is worth the added cost in terms of eradicating cancer, slowing its spread or improving patients’ lives, investors are reluctant to pony up money and insurance companies are reluctant to pay for treatments.
Studies that rigorously compare the results of proton or ion treatment with standard radiation therapy are just starting, says James Deye, program director for medical physics at the National Cancer Institute’s radiation research program.
The need for more research on ion therapy has caught the attention of the Department of Energy, whose Office of High Energy Physics oversees fundamental, long-term accelerator research in the US. A 2010 report, “Accelerators for America’s Future,” identified ion therapy as one of a number of areas where accelerator research and development could make important contributions to society.
In January 2013, more than 60 experts from the US, Japan and Europe met at a workshop sponsored by the DOE and NCI to identify areas where more research is needed on both the hardware and medical sides to develop the ion therapy systems of the future. Ideally, the participants concluded, future facilities should offer treatment with multiple types of charged particles—from protons to lithium, helium, boron and carbon ions—to allow researchers to compare their effectiveness and individual patients to get more than one type of treatment.
In June, the DOE’s Accelerator Stewardship program asked researchers to submit proposals for seed funding to improve accelerator and beam delivery systems for ion therapy.
“If there are accelerator technologies that can better enable this type of treatment, our job is to apply our R&D and technical skills to try to improve their ability to do so,” says Michael Zisman, an accelerator physicist from Lawrence Berkeley National Laboratory who is temporarily detailed to the DOE Office of High Energy Physics.
“Ideally we hope there will be partnerships between labs, industry, universities and medical facilities,” he says. “We don’t want good technology ideas in search of a problem. We rather want to make sure our customers are identifying real problems that we believe the application of improved accelerator technology can actually solve.”